RNA interference, or RNAi, is a natural biological process that inhibits the expression of a specific gene. In medicine, targeted RNAi therapies can be used to silence the expression of a disease-causing gene. To understand RNAi, you first have to understand RNA.
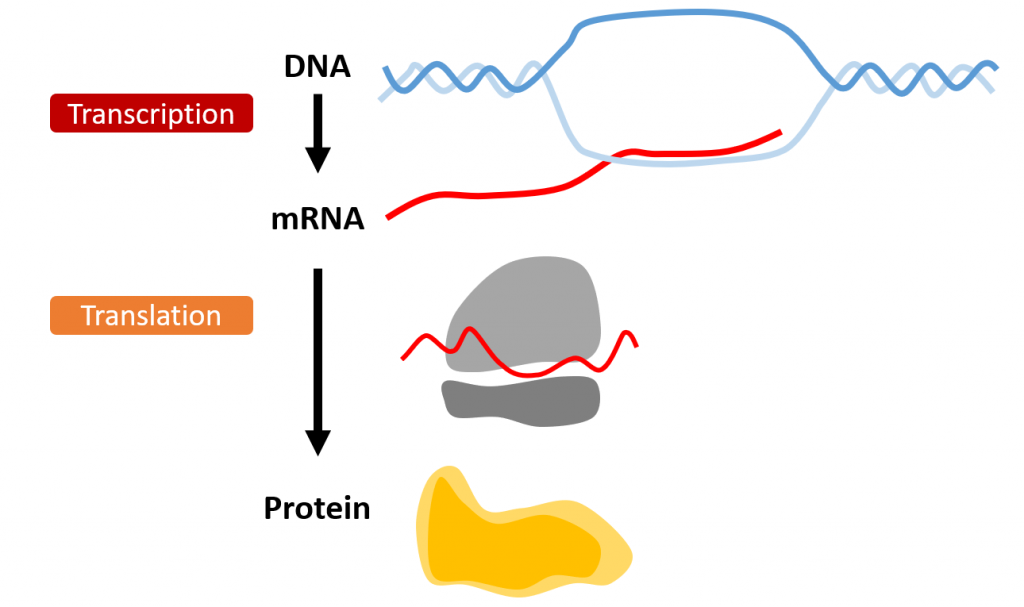
An overview of RNA is the messager between the DNA (the instructions) and the protein (the product). RNA is transcribed from the DNA. The ribosome translates the mRNA into protein. Graphic designed by Colleen Stoyas and illustrated by Celeste Suart.
Genes encode the instruction manual of our biology, but this material cannot leave the nucleus of your cells. Think of genes as a lecturer that provides instruction for your homework, which you must copy and take home to use later. The equivalent of copying this message in the cell is RNA, which transcribes the gene instructions and leaves the nucleus to be read and translated into protein. This protein then performs functions within the cell (see above image).
How can RNAi be used in ataxia?
In specific forms of ataxia, a gene mutation may provide the instructions for a protein that acts improperly and leads to disease. RNAi is a method of silencing RNA that interferes with the reading of this message, keeping a protein from being made. It works by generating a small interfering RNA in the laboratory that matches the gene of interest. When this small interfering RNA enters the cell, it binds the matching messenger RNA copied from a gene. When these two RNAs bind, the cell is triggered to cut up the message and destroy it. This means the disease-causing protein is never made. (see below image)
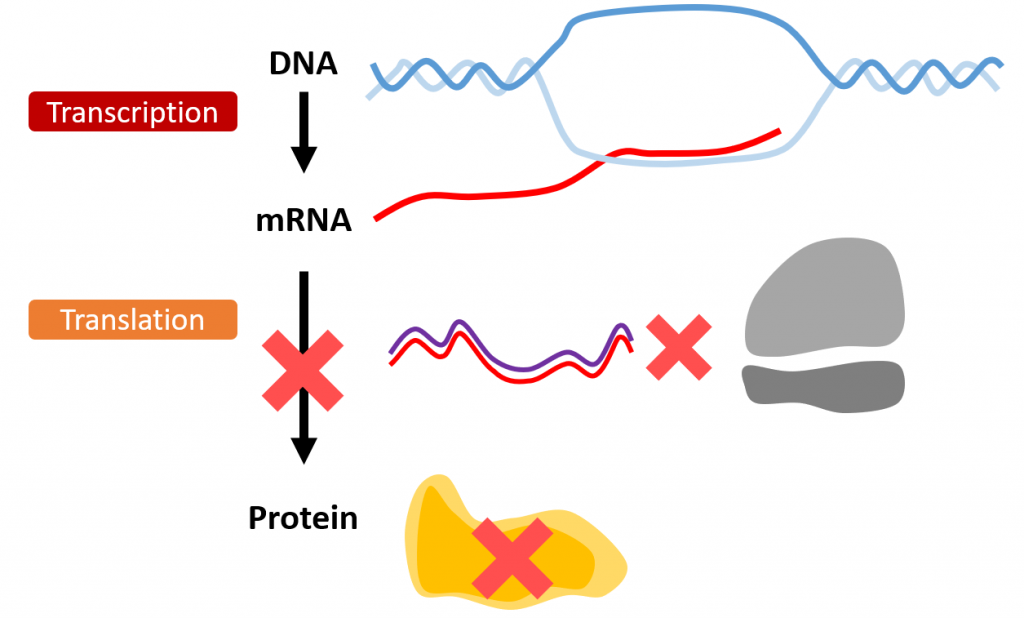
While RNAi is straightforward in the lab, getting it to work in humans can be tricky. The small interfering RNA cannot be taken in a pill, because it will not survive digestion. Additionally, the small interfering RNA is degraded along with the target messenger RNA, and so it must be continually administered. Using a viral payload, or encapsulating the interfering RNA in the coat proteins of a virus, has successfully delivered RNAi therapies in mouse models of SCA1, SCA3, and SCA7. In this method the virus integrates into your cells, which can then continue to produce the small interfering RNA. This means a single dose could potentially be all that is needed. Viral delivery to the brain is complicated, but not impossible. More work remains to be done clinically in order to determine if RNAi therapy is viable in a viral payload to treat multiple forms of spinocerebellar ataxia.
If you would like to learn more about RNAi, take a look at this video by TED-ED or entry in the Encyclopedia Britannica.
Snapshot written by Dr. Colleen Stoyas and edited by Frida Niss.
References
Costa Mdo C, Luna-Cancalon K, Fischer S, Ashraf NS, Ouyang M, Dharia RM et al (2013) Toward RNAi therapy for the polyglutamine disease Machado–Joseph disease. Mol Ther 21(10):1898–1908
Nobrega C, Nascimento-Ferreira I, Onofre I, Albuquerque D, Hirai H, Deglon N et al (2013) Silencing mutant ataxin-3 rescues motor deficits and neuropathology in Machado-Joseph disease transgenic mice. PLoS One 8(1):e52396
Ramachandran PS, Boudreau RL, Schaefer KA, La Spada AR, Davidson BL (2014) Nonallele specific silencing of ataxin-7 improves disease phenotypes in a mouse model of SCA7. Mol Ther 22(9):1635–1642
Scholefield J, Greenberg LJ, Weinberg MS, Arbuthnot PB, Abdelgany A, Wood MJ (2009) Design of RNAi hairpins for mutation-specific silencing of ataxin-7 and correction of a SCA7 phenotype. PLoS One 4(9):e7232’



