Written by Ambika Tewari Edited by Brenda Toscano Márquez
Trehalose, a natural autophagy inducer shows promise as a therapeutic candidate for MJD/SCA3
Every cell has an elaborate set of surveillance mechanisms to ensure optimal functioning. As proteins are synthesized, errors can occur leading to misfolded proteins. These abnormal proteins can be harmful to the cell. For this reasons it is important to monitortheir occurrence and decide whether they should be degraded. Autophagy is one way that these misfolded proteins can be degraded. Autophagy literally means self-eating and serves as a quality control mechanism. Defects in autophagy have been linked to several neurodegenerative disorders.
Machado-Joseph disease (MJD) or spinocerebellar ataxia type 3 is caused by an abnormal expanded CAG repeat in the ATXN3 gene. This CAG expansion causes misfolding of the ataxin-3 protein. The now unstable ataxin-3 is prone to forming aggregates in cells of some regions of the brain including the cerebellum, brainstem and basal ganglia. The accumulation of ataxin-3 in the cell leads to the progressive loss of neurons in the affected brain regions.
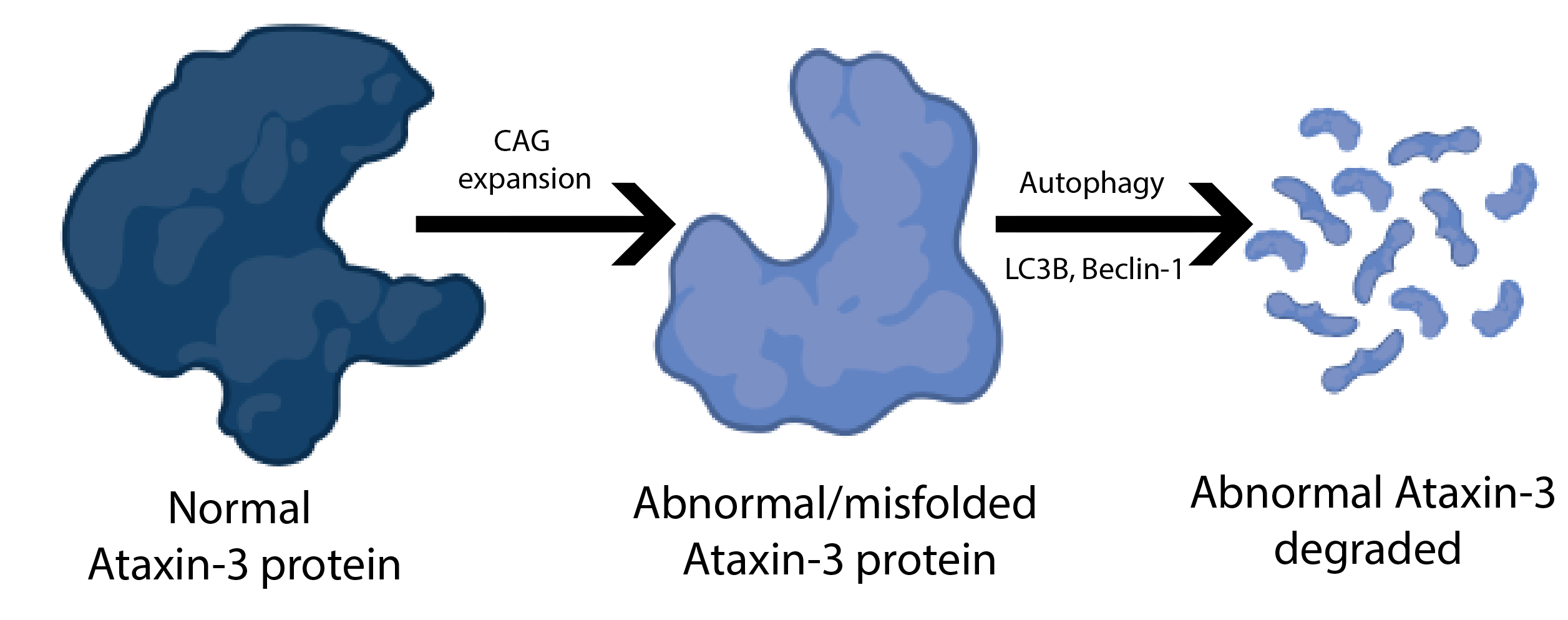
Researchers, eager to help patients with MJD, began to question why would the cellular surveillance system allow this toxic accumulation of misfolded ataxin-3. Surely there are mechanisms, like autophagy, to prevent this from occurring. This led to a number of studies that found that autophagy is defective in MJD patients. This was also confirmed in different mouse and cell models of MJD. In fact, earlier studies by the lab of Dr. Luís Pereira de Almeida found that increasing the amount of an autophagy protein (beclin-1) in the brain of an MJD mouse model improved some of the behavioral and neuropathological deficits. Together, these studies have provided evidence that autophagy may serve as a therapeutic target for MJD.
While restoring autophagy function in regions where it is defective is a worthwhile target for therapeutics, most of the tested methods involve invasive techniques to reach the brain and the use of viruses to deliver genes, which still needs to be optimized for use in human patients. In this study, the researchers identified a sugar additive, trehalose, which induces autophagy. Trehalose is approved for use in the food, cosmetic and pharmaceutical industries making it a readily-available agent for research studies.
To explore the efficacy of trehalose, researchers from the lab of Dr. Luís Pereira de Almeida began their studies in cell models. They expressed abnormal ataxin-3 (with 72 CAG repeats) in cultured cells and then added trehalose. By measuring the levels of LC3B, a protein that plays an important role in autophagy, they found that trehalose increased autophagy in cells 24 hours after treatment. Additionally, they saw a decrease in the protein levels of abnormal and endogenous ataxin-3. The promising results of these studies encouraged the researchers to test the effect of trehalose on mouse models of MJD.
The investigators used two different mouse models of MJD: a genetic mouse model (MJD-Tg) where animals express abnormal ataxin-3 since birth; and a lentiviral-based mouse model (MJD-lv) where abnormal ataxin-3 was injected into wild-type mice at 8 weeks of age. MJD-Tg mice express abnormal ataxin-3 only in Purkinje cells of the cerebellum, whereas MJD-lv mice express abnormal ataxin-3 in the striatum. Both groups of mice received an oral administration of 2% trehalose in their drinking water and they were compared to control mice who received water only.
Throughout the experiment, trehalose had no effect on the general health of the animals including their body weight. MJD-Tg mice were tested on the rotarod, which consists of a horizontally oriented rotating cylinder. The rotarod is a well-established test of motor dysfunction in mice. Treated mice showed improvement on the rotarod when rotating at constant speed but not on the accelerating rotarod, which requires highly coordinated movement. In both mouse models of MJD, there are prominent disease manifestations in the cerebellum or the striatum. Trehalose treatment decreased the size of mutant ataxin-3 aggregates in both models. In the cerebellum of MJD-Tg mice, the thickness of the granule cell layer was improved (i.e. increased in thickness) relative to untreated mice.
In summary, this study nicely used a range of different models of MJD to test the effects of trehalose on preventing or reversing the toxic accumulation of ataxin-3 aggregates. While trehalose seems like a plausible therapeutic candidate because of its safety profile and current approval across various industries, the preliminary data in this study did not show robust improvement in motor symptoms in the mouse models. It is challenging to find an ideal mouse model of a disorder. Unlike what happens in human patients, the two models of MJD used in this study were limited to a cell type or brain region. A more thorough follow-up study in a mouse that reflects a more systemic expression of abnormal ataxin-3 is warranted to determine the mechanism of action of trehalose. This could allow a more targeted approach for treatment with a better clinical outcome.
Key Terms
Misfolding: Proteins are folded into three dimensional structures. Mutations can cause abnormal folding, therefore changing the structure of a protein
Neuropathology: Pathological features of the nervous system associated with a disease, such as loss of neurons
Cell models: Individual cells grown together in a dish with special food and storage conditions to keep them alive throughout the entire length of an experiment
Endogenous: Natural or normally expressed ataxin-3, as opposed to ataxin-3 which is added into the system from external sources.
Genetic mouse model: A novel mouse that has its ataxin-3 gene altered using genetic engineering
Lentiviral-based mouse model: A normal mouse injected into a specific region of the brain with a virus that expresses ataxin-3
Conflict of Interest Statement
Ambika Tewari and Brenda Toscano Marquez have no conflicts of interest to declare.
Citation of Article Reviewed
Santana, M.M et al., Trehalose alleviates the phenotype of Machado-Joseph disease mouse models. J Transl Med, 2020. 18:161 (https://doi.org/10.1186/s12967-020-02302-2)



