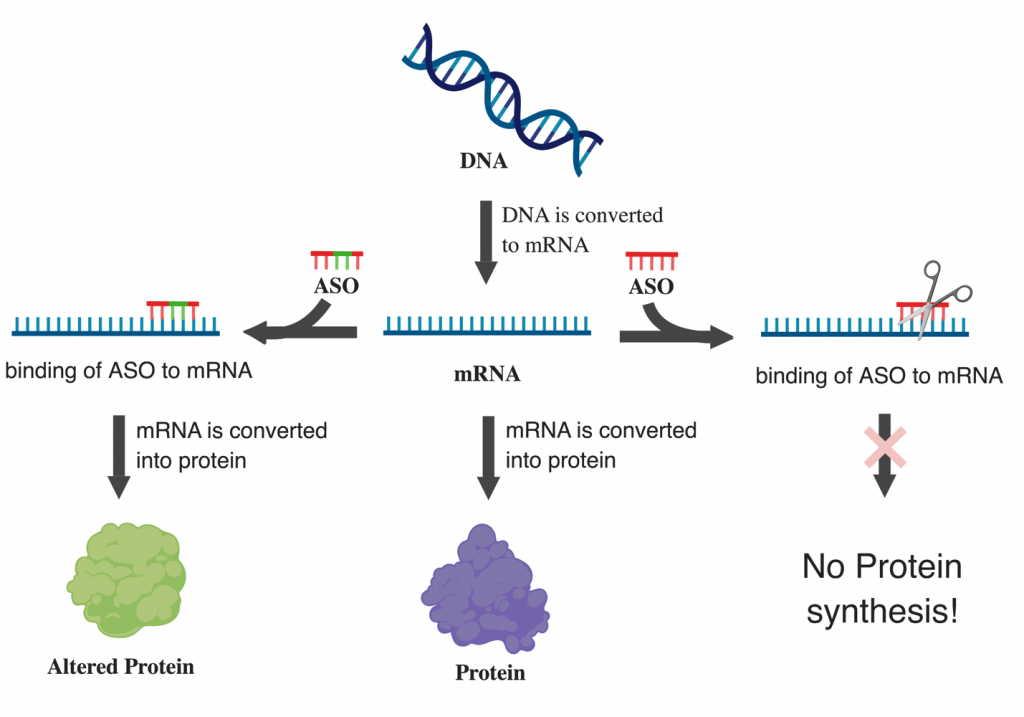
Antisense Oligonucleotides (also known as ASOs or AONs) are small molecules that can be used to prevent or alter the production of proteins. Proteins are the workforce of the cell, taking care of most cellular processes. They are generally made in a two-step process: first, a specific protein-coding gene is converted into an instruction file, called the messenger RNA (mRNA). The mRNA carries the information from that gene to the compartment of the cell that builds proteins. There, the mRNA’s information then gets converted into the protein. ASOs are short single stranded pieces of DNA that match the complementary sequence of a specific mRNA. Based on the type of chemical modifications, the ASO can have two different effects on the mRNA. Some modifications of ASOs trigger the destruction of the mRNA. This will result in the loss of the corresponding protein. Other modifications can mask only certain parts of the mRNA leading to a modified version of the protein.
The majority of Spinocerebellar Ataxias (SCAs) are caused by the accumulation of toxic proteins in certain regions of the brain. The primary goal of ASO treatments for SCAs is therefore to prevent the production of the toxic protein altogether. One example is work from Dr. Harry Orr’s group at the University of Minnesota. His lab studies Spinocerebellar Ataxia Type 1 (SCA1), which is caused by the toxic accumulation of the Atxn1 protein. Injections of ASOs into a SCA1 animal model decreased Atxn1 levels and rescued the SCA1 motor incoordination symptoms. Another way of using ASOs as treatment for SCAs is the modification of the mRNAs information to produce a modified version of the protein. This approach has been tested in Spinocerebellar Ataxia Type 3 (SCA3), in which an expansion in the Atxn3 gene renders the Atxn3 protein toxic. The van Roon-Mom group from the Netherlands, for instance, used ASOs to only remove the expansion from Atxn3 while leaving the remaining protein structure and function intact.
Both studies as well as other studies performed for additional SCAs are highlighting the potential use of ASOs as therapeutics for SCAs. While ASO research for SCA is mostly in the pre-clinical phase, ASO treatment for other diseases, including Duchenne muscular dystrophy and spinal muscular atrophy, have already gained approval by the US Food and Drug Administration (FDA). Further clinical trials will need to be performed to measure the therapeutic benefit of ASOs in SCA patients.
If you would like to learn more about antisense oligonucelotides, take a look at this article in HDBuzz about ASOs in development for Huntington’s Disease.
Snapshot written by Larissa Nitschke edited by Dr. Hayley McLoughlin



