Immunofluorescence is a common laboratory technique used to see proteins in a sample through microscopy. It relies on attaching a fluorophore, a chemical compound that can release light of a certain colour, to an antibody that can recognize a specific protein of interest.
Immunofluorescence is an important tool for studying biological processes at the cellular level. It can provide valuable information to researchers, such as the presence and the location of a specific protein within a cell. It is a simple yet robust method of studying a variety of samples, from single cells to complex tissues. Most importantly, it causes minimal disturbance to the shape and the surroundings of the protein of interest.
How does it work?
The technique utilizes the characteristic property of antibodies to bind to proteins of interest. A fluorophore, which emits specific wavelengths of light, is attached to the antibody. This combination of fluorophore and antibody lets us see the specific proteins under a microscope.
Different fluorophores can re-emit light at different wavelengths, thus producing different colours. Because of this, you can also simultaneously visualize multiple proteins.
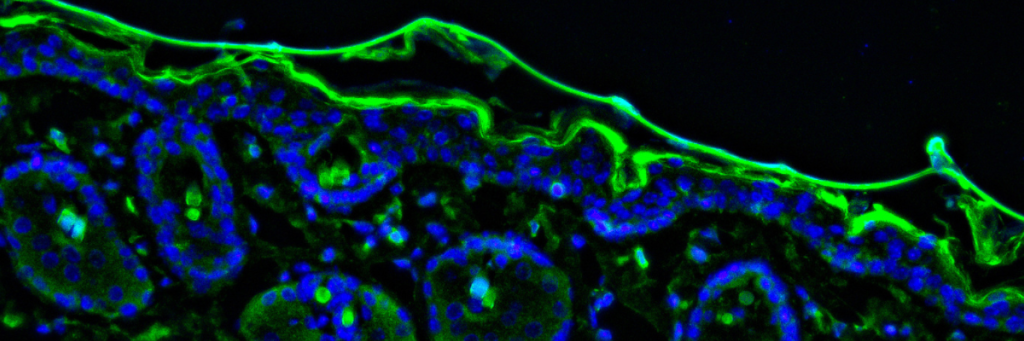
For instance, cellular structures such as the cytoskeleton can be labelled with a fluorophore emitting red light, while a protein of interest can be labelled with a fluorophore emitting green light to see if the protein localizes to the cytoskeleton (see figure 1). This information can further our understanding of how the protein works.
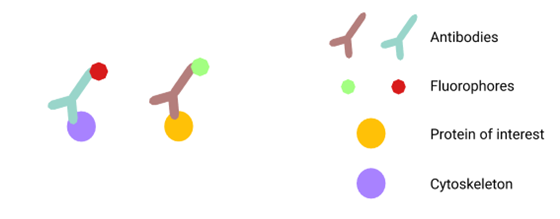
Figure 1. You can visualize multiple proteins at once using immunofluorescence, as long as you use different antibodies with fluorophores that are different colours. Figure by Christina (Yi) Peng.
Direct vs Indirect Immunofluorescence.
There are two types of immunofluorescence, direct and indirect (see figure 2).
In direct immunofluorescence, the antibody that recognizes the specific protein has a fluorophore attached. Thus, the fluorophore is directly connected to the protein.
In indirect immunofluorescence two antibodies are used. The primary antibody recognizes the protein, and a secondary antibody with a fluorophore attached recognizes the primary antibody. Thus, the fluorophore is indirectly connected to the protein. It is more complicated to perform due to the use of a secondary antibody, but it has the benefit of lower cost, broader applicability, and higher sensitivity.
It is more expensive to develop an antibody to use in direct immunofluorescence. The choice of fluorophore is also limited to one specific colour in direct immunofluorescence. Since you can only use one antibody of each colour per experiment, this limits the number of antibodies you can use in combination through direct immunofluorescence. For example, you couldn’t use two direct immunofluorescence antibodies with red labels in the same experiment.
Meanwhile, the antibodies for indirect immunofluorescence often are more inexpensive. The indirect method also allows multiple secondary antibodies, thus more fluorophores, to be attached to a single protein, amplifying signals that might have been too weak to detect otherwise.
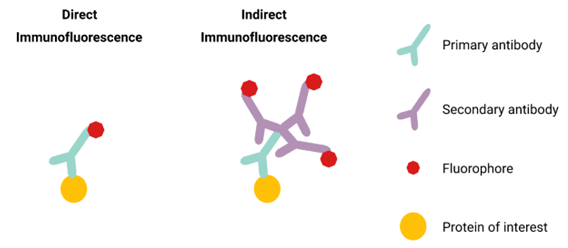
Figure 2. Direct vs Indirect Immunofluorescence. Figure by Christina (Yi) Peng.
If you would like to learn more about Immunofluorescence, take a look at these resources by Sajid Shaikh (YouTube) and the Addgene.
Snapshot Written by: Christina (Yi) Peng
Edited by: Dr. Larissa Nitschke

Snapshot: What Are Silent Mutations?
Our DNA (deoxyribonucleic acid) serves as a genetic blueprint for building and maintaining our tissues. This complex molecule contains the information needed to build proteins, written in a code made Read More…

Snapshot: What is a Variant of Uncertain Significance (VUS)?
What is a variant? The human genome contains over 3 billion base pairs– individual “letters” in the DNA code that make the instructions for our cells and bodies to function. Read More…

Snapshot: What Are Nonsense Mutations?
Our DNA (deoxyribonucleic acid) serves as a genetic blueprint for building and maintaining our tissues. This complex molecule contains the information needed to build proteins, written in a code made Read More…



