Written by Logan Morrison Edited by Dr. Sriram Jayabal
Stanford researchers accidentally discover a new role (reward prediction) for the cerebellum, the primary brain region affected by spinocerebellar ataxias.
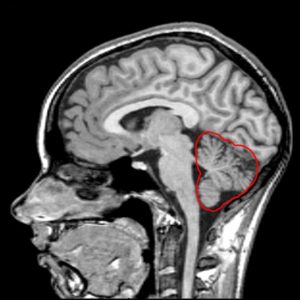
Would you believe that the part of your brain that enables you to perform simple, everyday tasks (like jogging or walking) also controls your ability to do more complex tasks (like throwing a curve ball) with accuracy? It’s true! Every one of our body’s movements is adjusted by a brain region known as the cerebellum – a primary area of pathology in spinocerebellar ataxias. The name “cerebellum” is a combination of the Latin word for the brain – cerebrum – and the Latin suffix -ellus, which means small. While this “little brain” might not take up much room, it actually contains the vast majority of the nerve cells (known as neurons) in the central nervous system1. Take a look at the image included with this article to see for yourself: even without the red highlighting, the cerebellum should be instantly recognizable as the distinctive structure in the bottom right, so folded and densely-packed that it looks a bit like something you’d find on the branches of a fern or shrub. Among these many folds are the circuits that fine-tune our motor output, providing us with the ability to move our bodies with ease and precision.
For decades, not much else was said about the function of the cerebellum beyond its primary role in tweaking movement. Recently, though, there have been some hints that there is more to this part of the brain than we might have thought: brain imaging studies of patients suffering from bipolar disorder, for instance, have sometimes shown abnormalities in the cerebellum3, 4. Cerebellar abnormalities have been implicated in a variety of other diseases, as well, including autism spectrum disorders, schizophrenia, Alzheimer’s disease, and multiple sclerosis5, 6. Now, thanks to the hard work of scientists at Stanford University7 – as well as a bit of luck – we know that the cerebellum is not only involved in how we move, but why.
Mark Wagner, a postdoctoral researcher in the lab of Liqun Luo, and Tony Kim, a graduate student in the lab of Mark Schnitzer, began their collaborative work with the intention of determining the exact processes that occur in the cells of the cerebellum when an animal makes certain movements. To accomplish this, they planned to use a cutting-edge technique known as two-photon calcium imaging to image the brains of awake mice while they performed movement-related tasks. In short, this involves mice that have been genetically engineered in such a way that their neurons emit fluorescent light whenever they absorb calcium (a crucial step for the communication between neurons). So, whenever one of these cells in the cerebellum becomes active, the electrical pulse it sends out is recorded as a flash of light. Then, in a brief surgical procedure, a small portion of the skull directly above the cerebellum is replaced with a thin glass pane. What makes this whole procedure so special is the hardware: the device that is subsequently used to peer through the glass pane and takes images is a two-photon microscope, which can generate images in real-time with such high resolution that we can make out individual cells much more clearly than ever before.
Wagner and Kim were interested in how the most abundant neurons of the cerebellum, known as granule cells, help mice execute precision arm movements. The only problem is that mice, left on their own to freely behave, do not typically move their front paws in the way the experimenters were interested in (at least, not in any consistent way). In order to address this, the researchers trained their mice to perform a simple lever-pulling task, in which mice would manipulate a small lever to receive a reward of sugar-water. Like us, mice love sugar, so each mouse practiced working the lever until they could reliably get their sweet treat. While they were repeatedly making the same arm movements, the two-photon microscope recorded the activity of the granule cells in their cerebellum.
As the researchers expected, different types of movement corresponded to activation in different groups of neurons. However, Wagner and Kim also noticed a distinct group of granule cells that became active whenever the mice were waiting for (that is, expecting) their reward. They determined that these neurons were not associated with motor function because they would fire no matter what movement was occurring – whether the mouse was pushing the lever forward, pulling the lever backward, or licking its lips in anticipation. It turned out that these “reward-anticipation cells,” as the researchers called them, would only stop firing once the mouse received their reward. Wagner and Kim even slipped in a few trials where they replaced the mouse’s reward with regular water, and in those cases, the reward-anticipation cells would continue to fire. In addition, they found that a separate group of granule cells only became active if (and only if) the mice received their reward. Without even meaning to, this team had discovered that the cerebellum is not just responsible for motor fine-tuning: it has a clear role in processing how we perceive our surroundings.
This landmark study suggests that the way our brains plan our movements takes into account not only the muscles we think we might need to flex, but also whether we believe the outcome of this movement will be rewarding. As we currently understand it, the cerebellum receives two basic types of information: what we plan to do when we make a movement, and the actual result of that movement (as seen by our eyes and felt by our skin/muscles). For example, let’s say we try to hold up two fingers on our right hand, but instead we extend all five. In that case, the cerebellum would detect a significant difference in our motor plan and our motor outcome. This discrepancy would be impossible to avoid in the future without the cerebellum, which is believed to correct such motor errors by sending highly-specific “teaching” signals to the other areas of the brain’s motor system8, 9. But if we believe a movement will result in a positive, rewarding experience, and then it turns out that it actually doesn’t, isn’t this also a form of error? Wagner and Kim’s work suggests that our brains might very well perceive it this way. More importantly, though, they have shown that this “motivation error” is processed, at least in part, in the same way that a motor error is: using the powerful circuits within the cerebellum.
What this means for SCA patients is that progression of the disease in the cerebellum might corrupt the brain’s motivation output in addition to its motor output. This has been hinted at recently in studies that have shown psychological symptoms such as mild depression and impaired decision-making in a subset of SCA patients10, 11. And now that we have a better understanding of what might be behind these symptoms, we are one step closer to figuring out how to effectively manage these kinds of cognitive deficits in the SCAs (though an assortment of drug trials in animals and clinical trials with patients would still be needed to determine whether any potential treatment could be viable). In addition, the high-profile nature of this discovery should help make it clear to everyone in the scientific and medical community – from doctors and clinical researchers to biologists and teachers across the world – that the cerebellum is more than just a cog in the motor system machine. Hopefully, by raising awareness of the cerebellum’s possible role in motivating our behavior, this research will draw more attention to the subtler psychological symptoms of SCAs that may have previously been overlooked.
If you would like to read more about this discovery, check out what the researchers have to say about it in Stanford’s recent press release.
Key Terms
Cerebellum: A primary area of pathology in the spinocerebellar ataxias. This brain region sits toward the back of the skull and, though small in stature, contains the majority of the nerve cells (neurons) in the central nervous system. Contains the circuits that fine-tune our movements, giving us the ability to move with precision.
Conflict of Interest Statement
The author and editor declare no conflicts of interests.
Citation of Article Reviewed
Wagner, M.J., Kim, T.H., Savall, J., Schnitzer, M.J. and Luo, L., Cerebellar granule cells encode the expectation of reward. Nature, 2018. 544(7648): p96. (https://www.ncbi.nlm.nih.gov/pubmed/28321129)
References
- Herculano-Houzel, S., The human brain in numbers: a linearly scaled-up primate brain. Front Hum Neurosci, 2009. 3: p. 31.
- Jensen, K. MRI Pictures. Central Nervous System – Visual Perspectives; Available from: http://cnsvp.stanford.edu/mri/index.html.
- Ketter, T.A., et al., Effects of mood and subtype on cerebral glucose metabolism in treatment-resistant bipolar disorder. Biol Psychiatry, 2001. 49(2): p. 97-109.
- Mills, N.P., et al., MRI analysis of cerebellar vermal abnormalities in bipolar disorder. Am J Psychiatry, 2005. 162(8): p. 1530-2.
- Mormina, E., et al., Cerebellum and neurodegenerative diseases: Beyond conventional magnetic resonance imaging. World J Radiol, 2017. 9(10): p. 371-388.
- Becker, E.B. and C.J. Stoodley, Autism spectrum disorder and the cerebellum. Int Rev Neurobiol, 2013. 113: p. 1-34.
- Wagner, M.J., et al., Cerebellar granule cells encode the expectation of reward. Nature, 2017. 544(7648): p. 96-100.
- Wolpert, D.M. and Z. Ghahramani, Computational principles of movement neuroscience. Nat Neurosci, 2000. 3 Suppl: p. 1212-7.
- Schultz, W. and A. Dickinson, Neuronal coding of prediction errors. Annu Rev Neurosci, 2000. 23: p. 473-500.
- Klinke, I., et al., Neuropsychological features of patients with spinocerebellar ataxia (SCA) types 1, 2, 3, and 6. Cerebellum, 2010. 9(3): p. 433-42.
- Ma, J., et al., Cognitive impairments in patients with spinocerebellar ataxia types 1, 2 and 3 are positively correlated to the clinical severity of ataxia symptoms. Int J Clin Exp Med, 2014. 7(12): p. 5765-71.



